Heat Capacity Formula
|
|
\( C \;=\; \dfrac{ \Delta Q }{ \Delta T }\) (Heat Capacity)
\( \Delta Q \;=\; C \cdot \Delta T \)
\( \Delta T \;=\; \dfrac{ \Delta Q }{ C }\)
|
| Symbol |
English |
Metric |
| \( C \) = Heat Capacity |
\(Btu\;/\;F\) |
\(kJ\;/\;K\) |
| \( \Delta Q \) = Heat Transfered Amount |
\(Btu\;/\;hr\) |
\( W \) |
| \( \Delta T \) = Temperature Differential |
\( F \) |
\( K \) |
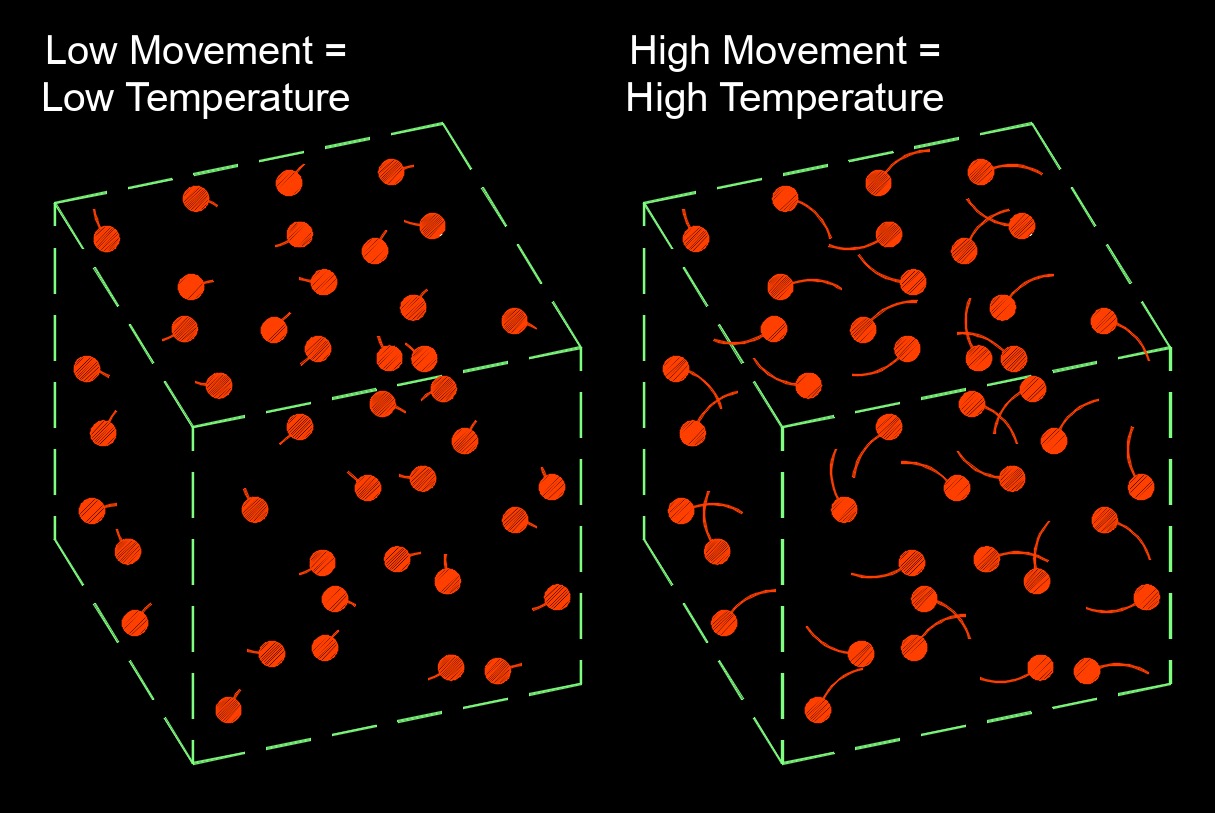 Heat capacity, abbreviated as C or \(c_p\), is the amount of enerigy required to increase the temperature of a substance by 1°C. The heat gain or loss results in a change in temperature and the state and performance of work. The heat capacity of a substance is an important property in thermodynamics as it helps to determine the amount of energy needed to heat or cool a substance or system. It also influences the rate at which a substance changes temperature in response to thermal energy transfer.
Heat capacity, abbreviated as C or \(c_p\), is the amount of enerigy required to increase the temperature of a substance by 1°C. The heat gain or loss results in a change in temperature and the state and performance of work. The heat capacity of a substance is an important property in thermodynamics as it helps to determine the amount of energy needed to heat or cool a substance or system. It also influences the rate at which a substance changes temperature in response to thermal energy transfer.
Different substances and systems have different heat capacities due to variations in their molecular structure and composition. The heat capacity of a substance may also vary with temperature, particularly for materials that undergo phase transitions or exhibit other complex thermal behavior.


 Heat capacity, abbreviated as C or \(c_p\), is the amount of
Heat capacity, abbreviated as C or \(c_p\), is the amount of 