First Law of Thermodynamics
First Law of Thermodynamics formula
|
||
|
\( \Delta U \;=\; Q - W \) (First Law of Thermodynamics) \( Q \;=\; \Delta U + W \) \( W \;=\; Q - \Delta U \) |
||
| Symbol | English | Metric |
| \(\triangle U\) = internal energy | \(lbf-ft\) | \(J\) |
| \(Q\) = heat added to the system | \(^\circ F\) | \(^\circ C\) |
| \(W\) = work done by the system | \(lbf-ft\) | \(J\) |
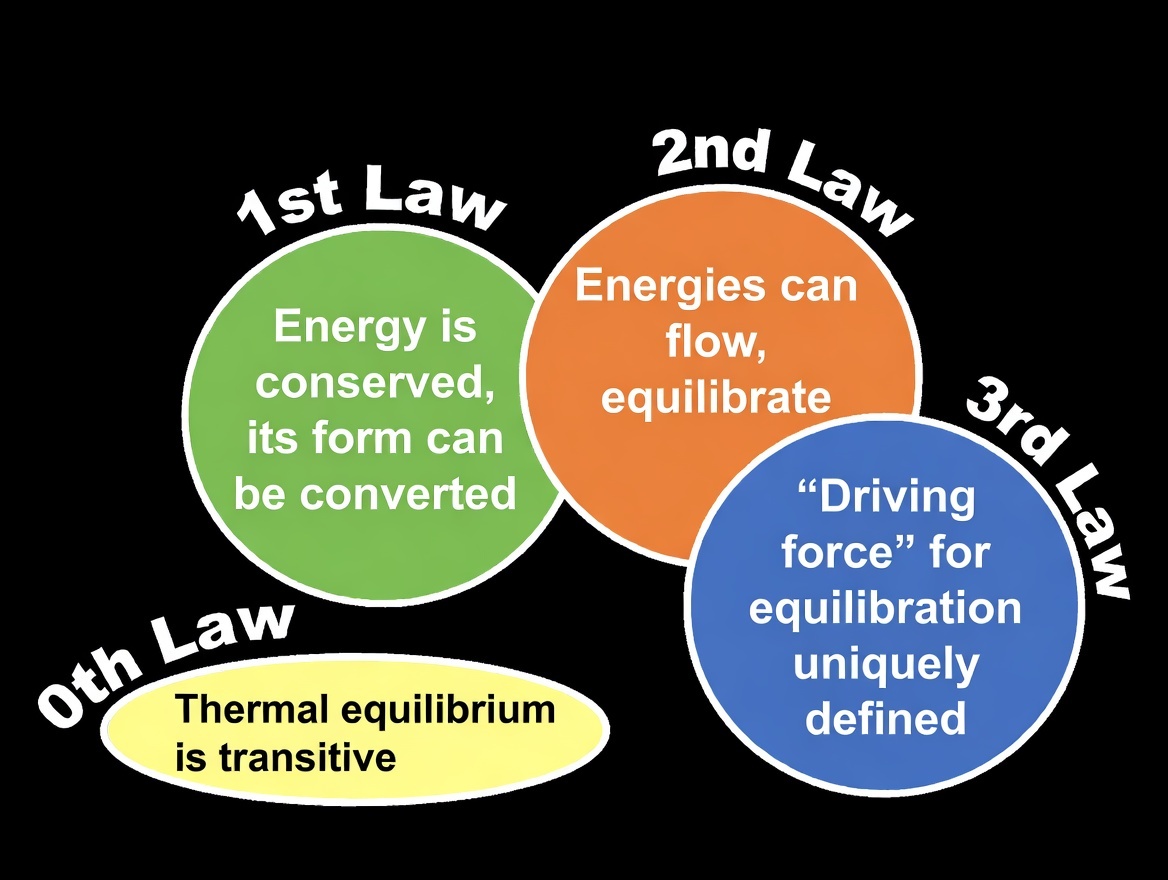 First law of thermodynamics, also called law of conservation of energy, states that the total energy of an isolated system remains constant. This means that energy can neither be created nor destroyed but can only be transferred or converted from oneform to another. For a closed system, the change in internal energy \((\Delta U)\) is equal to the heat added to the system \((Q)\) minus the work done by the system on its surroundings \((W)\), expressed mathematically as \( \Delta U = Q - W \). This principle is foundational in fields like mechanical engineering, where it governs the analysis of combustion engines, refrigeration cycles, heat exchangers, and power plants, ensuring that energy balances are maintained in system designs.
First law of thermodynamics, also called law of conservation of energy, states that the total energy of an isolated system remains constant. This means that energy can neither be created nor destroyed but can only be transferred or converted from oneform to another. For a closed system, the change in internal energy \((\Delta U)\) is equal to the heat added to the system \((Q)\) minus the work done by the system on its surroundings \((W)\), expressed mathematically as \( \Delta U = Q - W \). This principle is foundational in fields like mechanical engineering, where it governs the analysis of combustion engines, refrigeration cycles, heat exchangers, and power plants, ensuring that energy balances are maintained in system designs.
In practical applications, engineers apply the first law to control volumes or open systems, where mass can enter or exit, leading to the steady flow energy equation. This accounts for enthalpy changes, kinetic and potential energy, in addition to heat and work transfers. For instance, in a turbine, the energy extracted as shaft work comes from the decrease in the fluid's enthalpy, with heat losses considered to verify efficiency.

