Avogadro's Law
Avogadro's law, also called Avogadro's principle, one of the Gas Laws, when temperature and pressure are held constant, states that, at the same temperature and pressure, equal volumes of different gases contain the same number of molecules.
Avogadro's Law Formula |
||
|
\( \dfrac{ V_1 }{ n_1 } \;=\; \dfrac{ V_2 }{ n_2 } \) (Avogadro's Law) \( V_1 \;=\; \dfrac{ n_1 \cdot V_2 }{ n_2 } \) \( n_1 \;=\; \dfrac{ V_1 \cdot n_2 }{ V_2 } \) \( V_2 \;=\; \dfrac{ V_1 \cdot n_2 }{ n_1 } \) \( n_2 \;=\; \dfrac{ n_1 \cdot V_2 }{ V_1 } \) |
||
| Symbol | English | Metric |
| \( V_1 \) = Initial Volume of Gas | \( in^3 \) | \(mm^3 \) |
| \( n_1 \) = Initial Moles of Gas | \(mol\) | \(mol\) |
| \( V_2 \) = Final Volume of Gas | \( in^3 \) | \( mm^3 \) |
| \( n_2 \) = Final Moles of Gas | \(mol\) | \(mol\) |
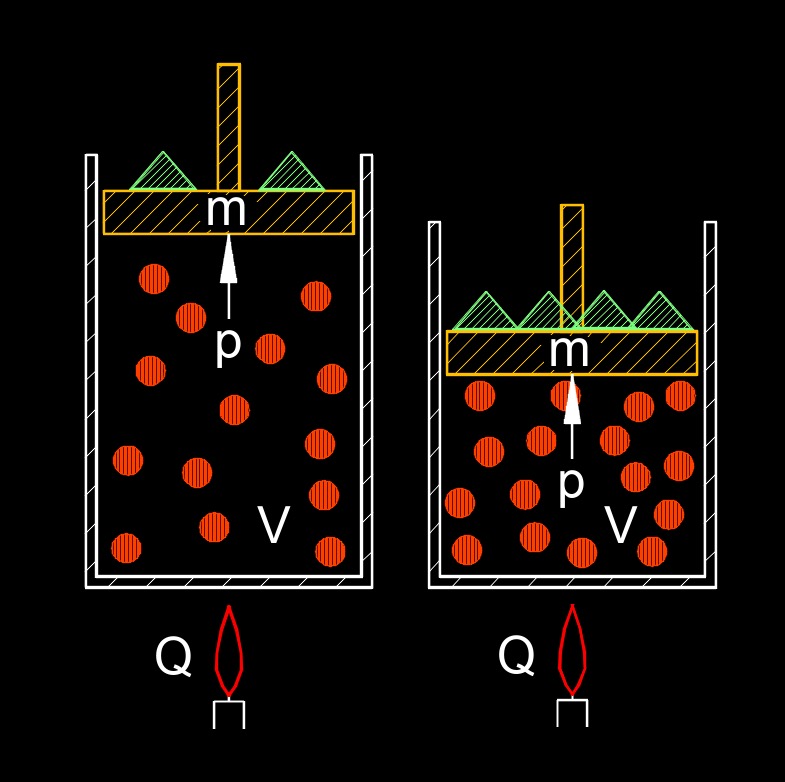 This law shows that the ratio of the volume of a gas to the number of moles of the gas is constant, as long as the temperature and pressure remain constant. This means that if the number of moles of a gas is doubled, the volume of the gas will also double, as long as the temperature and pressure remain constant. This law is an important concept in chemistry and physics, and is used to explain the behavior of gases and the properties of materials.
This law shows that the ratio of the volume of a gas to the number of moles of the gas is constant, as long as the temperature and pressure remain constant. This means that if the number of moles of a gas is doubled, the volume of the gas will also double, as long as the temperature and pressure remain constant. This law is an important concept in chemistry and physics, and is used to explain the behavior of gases and the properties of materials.

