Evaporation
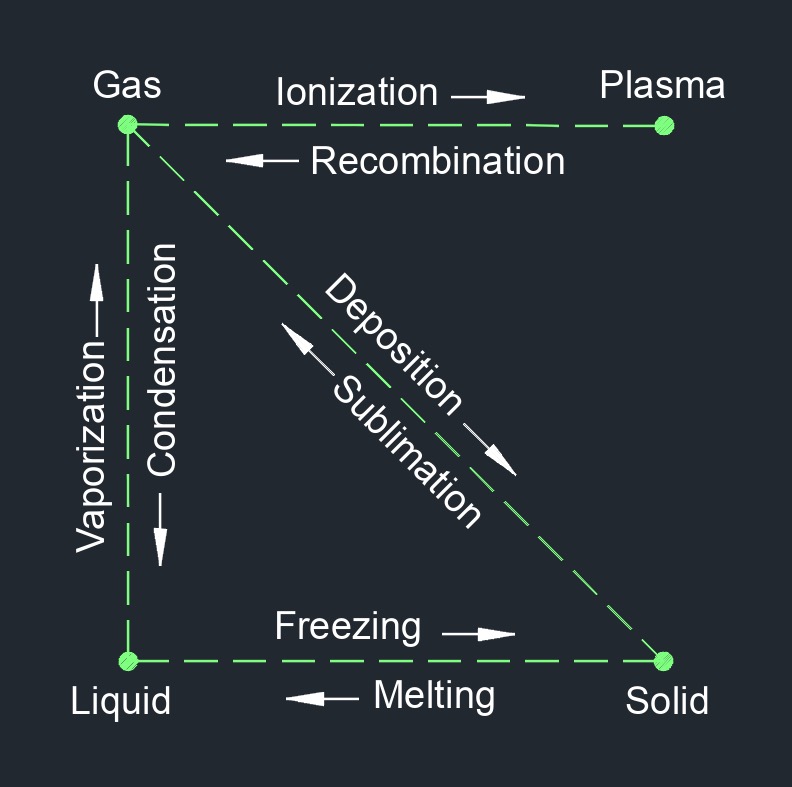
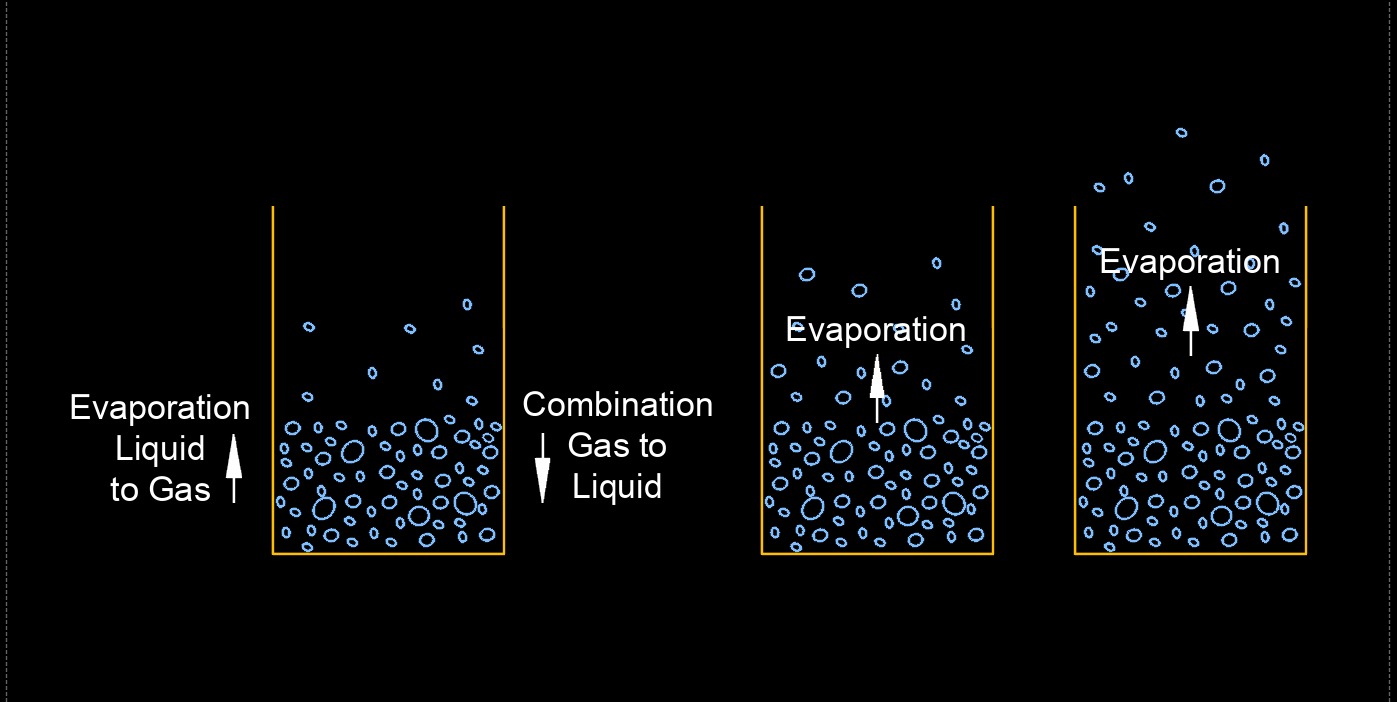 Evaporation is the process by which a liquid, such as water, changes from a liquid state to a gaseous state. This occurs when the molecules of the liquid gain enough energy to overcome the intermolecular forces holding them together, allowing them to escape into the surrounding environment as vapor or gas. The rate of evaporation is influenced by several factors, including the temperature of the liquid, the surface area of the liquid, the humidity of the surrounding air, and the presence of other substances in the liquid or in the surrounding environment.
Evaporation is the process by which a liquid, such as water, changes from a liquid state to a gaseous state. This occurs when the molecules of the liquid gain enough energy to overcome the intermolecular forces holding them together, allowing them to escape into the surrounding environment as vapor or gas. The rate of evaporation is influenced by several factors, including the temperature of the liquid, the surface area of the liquid, the humidity of the surrounding air, and the presence of other substances in the liquid or in the surrounding environment.
Evaporation is an important process in the water cycle, as it contributes to the formation of clouds and precipitation. It is also important in many industrial processes, such as drying, distillation, and desalination. Evaporation can be enhanced through the use of various methods such as increasing the temperature or airflow over the surface of the liquid, or reducing the pressure above the liquid. Conversely, evaporation can be inhibited through methods such as reducing the temperature, increasing the humidity of the surrounding air, or covering the liquid surface to reduce exposure to air.

