Liquid
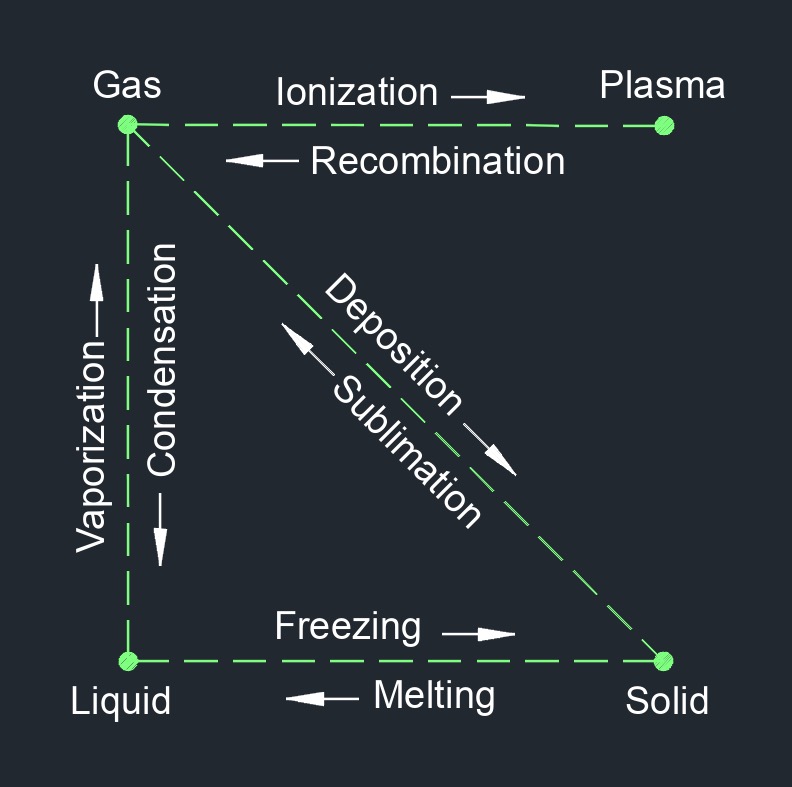
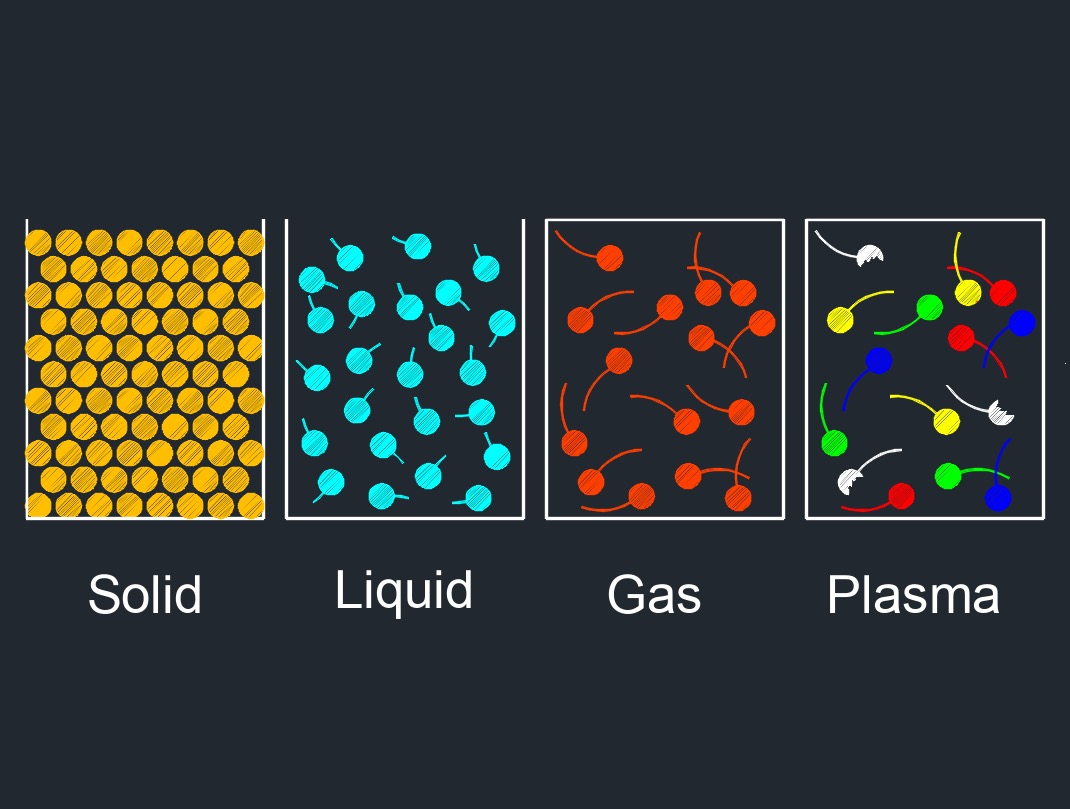 Liquid, abbreviated as \(LIQ\), has a specific volume and can hold any shape it is contained within. Depending on its elements, the density and weight will vary. A liquids molecules are close together with no particular pattern and continually move past each other, this allows it to flow and move about. Having little space between the molecules leaves little room to compress.
Liquid, abbreviated as \(LIQ\), has a specific volume and can hold any shape it is contained within. Depending on its elements, the density and weight will vary. A liquids molecules are close together with no particular pattern and continually move past each other, this allows it to flow and move about. Having little space between the molecules leaves little room to compress.
Liquid is one of the three common states of matter, along with solid and gas. In its liquid state, matter has a definite volume but no fixed shape. Liquids take the shape of their container, and they flow and move around freely, though they do not easily compress. The particles that make up a liquid are in constant motion and are loosely arranged, with weaker intermolecular forces than in solids. The molecules or atoms that make up a liquid are able to slide past each other, but are still closely packed together.
Liquid Properties |
|||
| Properties | Gas | Liquid | Solid |
| Compressibility | Yes | Low | No |
| Density | Low | Moderate | High |
| Expandability | Yes | Yes | No |
| Intermolecule force strength | Weak | Moderate | Strong |
| Particle Movement | Free movement | Free movement | No free movement |
| Shape | Infinite | Infinite | Fixed |
| Shear Resistance | Yes | Yes | No |
| Viscosity | Low | Varies | No |
| Volume | Infinite | Fixed | Fixed |
The properties of liquids, such as their boiling point, viscosity, and surface tension, depend on the behavior of the particles that make up the liquid. For example, liquids with strong intermolecular forces, such as water, have high surface tension and can form droplets, while liquids with weaker intermolecular forces, such as alcohol, have low surface tension and can spread out more easily.
Liquids are an important substance in many areas of science and technology, including chemistry, physics, engineering, and biology. Many everyday objects, such as drinks, cleaning products, and lubricants, are liquids. The properties of liquids are also important in understanding the behavior of materials under different conditions, such as at different temperatures and pressures.

