Cathodic Protection
Cathodic Protection, Electrical, Corrosion, Glossary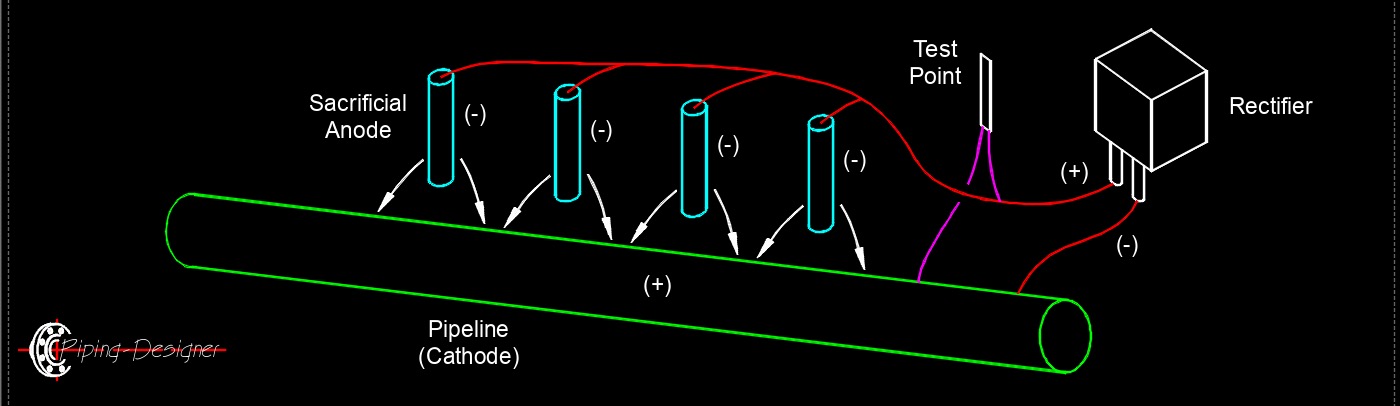 Cathodic protection, abbreviated as \(CP\), is a technique used to prevent corrosion of metal structures and equipment by making them the cathode of an electrochemical cell. Corrosion occurs when metal is exposed to an electrolyte, such as water or soil, and a flow of electrons occurs between the metal and the electrolyte, resulting in the metal corroding. By making the metal structure the cathode of an electrochemical cell, the flow of electrons is reversed, and corrosion is prevented. CP is typically achieved by connecting a sacrificial anode made of a more active metal, such as zinc or magnesium, to the metal structure being protected. The sacrificial anode corrodes instead of the metal structure, providing cathodic protection. Another method of cathodic protection is impressed current cathodic protection, which involves using a direct current power source to provide the required current flow to the structure being protected.
Cathodic protection, abbreviated as \(CP\), is a technique used to prevent corrosion of metal structures and equipment by making them the cathode of an electrochemical cell. Corrosion occurs when metal is exposed to an electrolyte, such as water or soil, and a flow of electrons occurs between the metal and the electrolyte, resulting in the metal corroding. By making the metal structure the cathode of an electrochemical cell, the flow of electrons is reversed, and corrosion is prevented. CP is typically achieved by connecting a sacrificial anode made of a more active metal, such as zinc or magnesium, to the metal structure being protected. The sacrificial anode corrodes instead of the metal structure, providing cathodic protection. Another method of cathodic protection is impressed current cathodic protection, which involves using a direct current power source to provide the required current flow to the structure being protected.
| Science |
| Applied Science |
| Engineering |
| Chemical Engineering |
| Material Science |
| Corrosion Engineering |
| Cathodic Protection |
CP is commonly used in a variety of industries, including oil and gas, marine, and transportation, to protect metal structures such as pipelines, tanks, and ships from corrosion. It is a highly effective technique for preventing corrosion, and can extend the life of metal structures significantly. CP systems require regular monitoring and maintenance to ensure that they continue to function properly over time. Corrosion engineers and technicians are responsible for designing, installing, and maintaining cathodic protection systems to ensure that metal structures remain protected from corrosion.
How Cathodic Protection Works
Cathodic protection is an electrochemical corrosion control technique used to prevent the deterioration of metallic structures such as pipelines, storage tanks, ship hulls, and reinforced concrete components. The method works by forcing the metal structure that requires protection to act as a cathode in an electrochemical cell. Because corrosion is fundamentally an oxidation reaction in which metal atoms lose electrons, converting the structure into a cathode suppresses this oxidation reaction and significantly reduces or stops corrosion.
There are two widely used methods of cathodic protection. The first is sacrificial anode cathodic protection, also called the galvanic anode method. In this approach, a more electrochemically active metal, commonly magnesium, zinc, or aluminum, is electrically connected to the structure being protected. These metals have a more negative electrode potential and therefore preferentially corrode. As the sacrificial anode oxidizes, it releases electrons that flow to the protected structure, keeping the structure in a cathodic state and preventing its metal from oxidizing.
The second method is impressed current cathodic protection (ICCP). In this system, an external direct current power supply forces electrical current from inert or semi-inert anodes (often graphite, mixed metal oxide, or high-silicon cast iron) through the electrolyte, such as soil or water, to the structure being protected. The external power source maintains the structure at a sufficiently negative electrical potential so that corrosion reactions on the protected metal surface are suppressed.
Both methods rely on the same fundamental principle of electrochemistry, corrosion occurs at anodic sites where metal oxidation takes place, and by supplying electrons to the structure, cathodic protection eliminates or greatly reduces the formation of these anodic corrosion reactions. Proper design, monitoring of structure-to-electrolyte potential, and maintenance are necessary to ensure that the system provides adequate protection over time.
Cathodic Protection Anode Types

