Solid
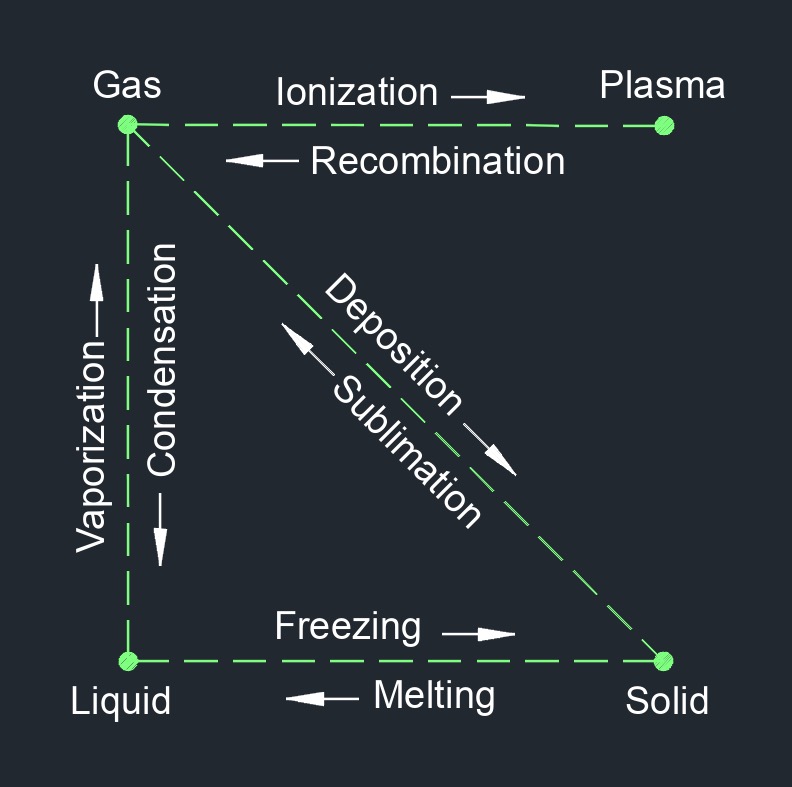
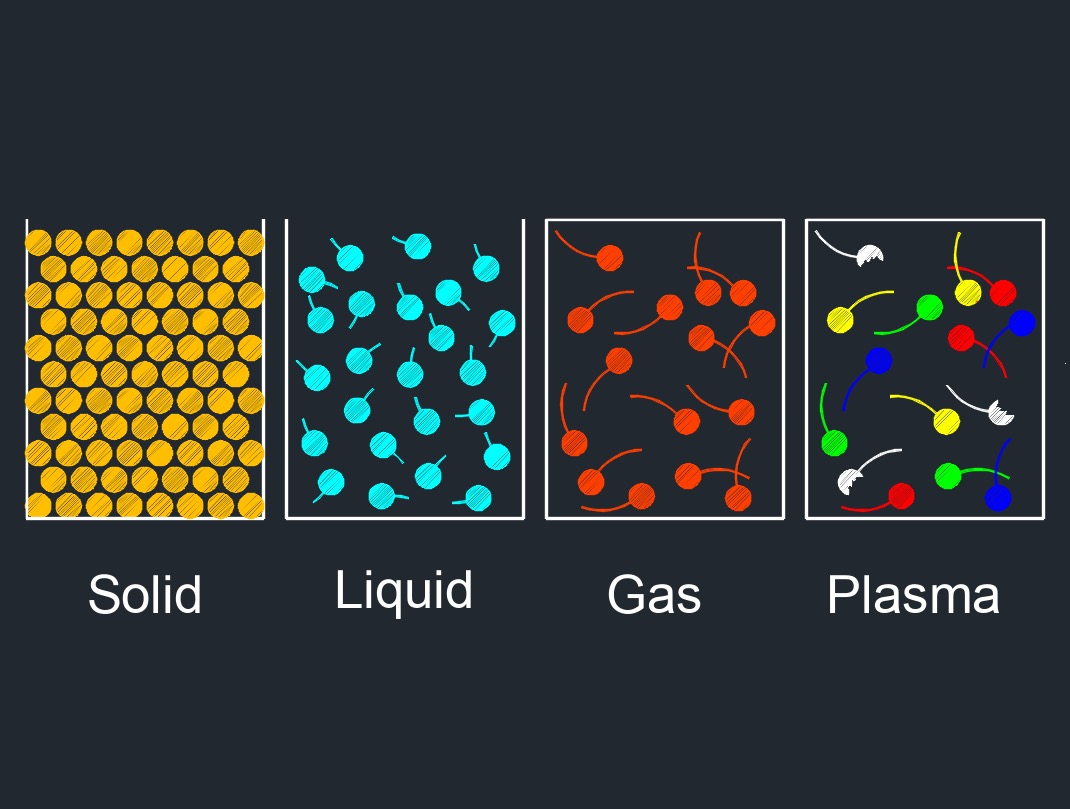 A solid has particles that are compressed together in an orderly pattern. Solid are one of the three common states of matter, along with liquid and gas. In its solid state, matter has a definite shape and volume, and the particles that make up the solid are tightly packed together in a fixed arrangement. Solids are not easily compressed and have a relatively high density compared to liquids and gases.
A solid has particles that are compressed together in an orderly pattern. Solid are one of the three common states of matter, along with liquid and gas. In its solid state, matter has a definite shape and volume, and the particles that make up the solid are tightly packed together in a fixed arrangement. Solids are not easily compressed and have a relatively high density compared to liquids and gases.
The properties of solids, such as their melting point, boiling point, and strength, depend on the arrangement and behavior of the particles that make up the solid. In a solid, the particles vibrate around their fixed positions, but do not move around freely as they do in a liquid or gas.
Solid Properties |
|||
| Properties | Gas | Liquid | Solid |
| Compressibility | Yes | Low | No |
| Density | Low | Moderate | High |
| Expandability | Yes | Yes | No |
| Intermolecule force strength | Weak | Moderate | Strong |
| Particle Movement | Free movement | Free movement | No free movement |
| Shape | Infinite | Infinite | Fixed |
| Shear Resistance | Yes | Yes | No |
| Viscosity | Low | Varies | No |
| Volume | Infinite | Fixed | Fixed |
Solids are an important substance in many areas of science and technology, including materials science, engineering, and geology. Many everyday objects, such as furniture, tools, and electronics, are made from solids. The properties of solids are also important in understanding the behavior of materials under stress or in extreme conditions, such as at high temperatures or pressures.

